Classification
Labeling Chemistry
Introduction
Protein labeling reagents are used for various assays that are coupled with antigen-antibody reaction. Of these, fluorescent, biotin, and enzyme labeling reagents are used most frequently for antibody labeling. Most protein labeling reagents have similar reactive sites, such as succinimidyl ester (NHS) for amino groups and maleimide or bromoacetamide for sulfhydryl groups. Dojindo offers the following protein labeling reagents and kits for protein research and proteomics.
There are several reactive sites on proteins as indicated in Fig. 1. The most common reactive site used is the NH2 group from lysine residue because of the simple process of labeling an NH2 terminal under physiological conditions. Sometimes, protein labeling with NH2-reactive compounds is not favorable because of the potential blockage of the protein’s active site. However, activity loss due to blockage of active sites by labeling is rare in the case of antibody labeling with NH2-reactive reagent. Therefore, labeling at the NH2 group on a protein is fairly reliable and simple method for preparing a conjugate. The second most common labeling method is the use of SH-reactive compounds. Most proteins do not have an SH group, just a disulfide group. Disulfide groups can be converted to SH groups by reducing agents such as dithiothreitol (DTT) or β mercaptoethanol. Then, SH-reactive compounds such as maleimide and bromoacetyl compounds are applied in labeling the protein’s SH groups. Because the location of the disulfide group on an antibody is more specific than that of an NH2 group, activity loss by SH group labeling is minimal. The reduction process may cause activity loss of an antibody because of possible cleavage between a heavy chain and a light chain. However, a conjugate prepared by SH labeling provides a better signal than that prepared by NH2 labeling because of site-specific labeling. Overall, a conjugate prepared by NH2 labeling is adequate for general assays while a conjugate prepared by SH labeling can be used for high-sensitivity assays. The sugar chain of a protein is also a possible labeling site with hydrazide compounds. To convert
to an aldehyde group before labeling with a hydrazide labeling.
|
Fig. 1 Reaction site for protein labeling |
Labeling Kits
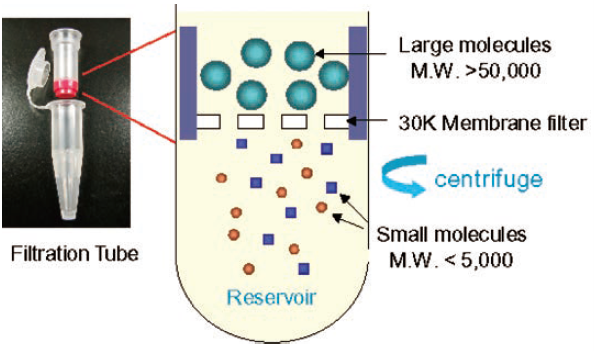
Dojindo offers a wide variety of protein labeling kits. These kits can be used to prepare conjugates with enzyme, fluorophore, and biotin (Table 1). There are two types of kits available: NH2 labeling (Kit-NH2) and SH labeling (Kit-SH). Dojindo’s labeling kits contain centrifugal Filtration Tubes that are used for protein purification, conjugation reaction, condensation, and buffer exchange. This single Filtration Tube format is the most unique feature of the kit (Fig. 2). Labeling requires 1) purification of protein and buffer exchange, 2) labeling reaction, and 3) purification and recovery of the conjugate. Because these three processes can be performed in a single Filtration Tube, recovery of conjugated proteins is always high, nearly 100% of protein can be recovered from the tube, and reproducibility is high as well. The Filtration Tube has a membrane that allows smaller molecules to pass through while larger molecules remain on the membrane. Through this simple centrifuge process, small proteins can be removed, leaving only conjugated proteins to be recovered by pippetting with a buffer solution.
|
Fig. 2 Filtration Tube with a 30K membrane filter and its molecule separation mechanism. |
Additionally, the kit’s unique buffer system prevents aggregation of protein and conjugate during labeling and storage of the conjugate. The conjugate recovered with the buffer (storage buffer) is stable for several months at 4°C or several years if it is stored at -20°C. A 10K membrane is available for proteins smaller than 50K. The enzyme labeling kits can be used for labeling small molecules, such as organic chemicals, amino acids, peptides, or oligonucleotides.
For more detailed information, please review the product information section. Kit-NH2 and Kit-SH include buffer solutions for washing, labeling reaction, and recovery of proteins; activated compounds; 30K Filtration Tubes; and an easy-to-follow protocol with photos. Kit-SH additionally contains reducing agents for converting disulfide groups to sulfhydryl groups (SH). The overall protocols for Kit-NH2 and Kit-SH for labeling macromolecules and small molecules g are indicated in Figs. 3 and 4. Most small molecules, such as buffer components, sodium azide (preservative), amine compounds, thiol compounds, detergents, glycerol, etc., are removed from proteins by the first step. Then, the appropriate labeling reagent is added directly to the Filtration Tube and incubated at 37°C for a certain time period. The conjugate is then recovered with storage buffer or WS buffer. The average concentration of protein will be 500-1,000 μg per ml as antibody. In most cases, the conjugates can be applied for downstream experiments such as ELISA, western blotting, tissue staining, and flowcytometry without further purification.
| Table 1 Labeling Kits | ||
| Category | Labeling Material | Target Group |
|---|---|---|
| Biotin Labeling | Biotin | amine, thiol |
| Enzyme Labeling | Peroxidase | amine, thiol |
| Alkaline phosphatase | amine, thiol | |
| Fluorophore Labeling | Phycobiliproteins | amine, thiol |
| Fluorescent dyes | amine, thiol | |
|
Fig. 3 Macromolecule labeling processes |
|
Fig. 4 Small molecule labeling processes |
Dojindo offers a wide variety of protein labeling kits. These kits can be used to prepare conjugates with enzyme, fluorophore, and biotin (Table 1). There are two types of kits available: NH2 labeling (Kit-NH2) and SH labeling (Kit-SH). Dojindo’s labeling kits contain centrifugal Filtration Tubes that are used for protein purification, conjugation reaction, condensation, and buffer exchange. This single Filtration Tube format is the most unique feature of the kit (Fig. 2). Labeling requires 1) purification of protein and buffer exchange, 2) labeling reaction, and 3) purification and recovery of the conjugate. Because these three processes can be performed in a single Filtration Tube, recovery of conjugated proteins is always high, nearly 100% of protein can be recovered from the tube, and reproducibility is high as well. The Filtration Tube has a membrane that allows smaller molecules to pass through while larger molecules remain on the membrane. Through this simple centrifuge process, small proteins can be removed, leaving only conjugated proteins to be recovered by pippetting with a buffer solution.
|
Fig. 2 Filtration Tube with a 30K membrane filter and its molecule separation mechanism. |
Additionally, the kit’s unique buffer system prevents aggregation of protein and conjugate during labeling and storage of the conjugate. The conjugate recovered with the buffer (storage buffer) is stable for several months at 4°C or several years if it is stored at -20°C. A 10K membrane is available for proteins smaller than 50K. The enzyme labeling kits can be used for labeling small molecules, such as organic chemicals, amino acids, peptides, or oligonucleotides.
For more detailed information, please review the product information section. Kit-NH2 and Kit-SH include buffer solutions for washing, labeling reaction, and recovery of proteins; activated compounds; 30K Filtration Tubes; and an easy-to-follow protocol with photos. Kit-SH additionally contains reducing agents for converting disulfide groups to sulfhydryl groups (SH). The overall protocols for Kit-NH2 and Kit-SH for labeling macromolecules and small molecules g are indicated in Figs. 3 and 4. Most small molecules, such as buffer components, sodium azide (preservative), amine compounds, thiol compounds, detergents, glycerol, etc., are removed from proteins by the first step. Then, the appropriate labeling reagent is added directly to the Filtration Tube and incubated at 37°C for a certain time period. The conjugate is then recovered with storage buffer or WS buffer. The average concentration of protein will be 500-1,000 μg per ml as antibody. In most cases, the conjugates can be applied for downstream experiments such as ELISA, western blotting, tissue staining, and flowcytometry without further purification.
| Table 1 Labeling Kits | ||
| Category | Labeling Material | Target Group |
|---|---|---|
| Biotin Labeling | Biotin | amine, thiol |
| Enzyme Labeling | Peroxidase | amine, thiol |
| Alkaline phosphatase | amine, thiol | |
| Fluorophore Labeling | Phycobiliproteins | amine, thiol |
| Fluorescent dyes | amine, thiol | |
|
Fig. 3 Macromolecule labeling processes |
|
Fig. 4 Small molecule labeling processes |
Enzyme Labeling Kits
Peroxidase and alkaline phosphatase are the most frequently used enzymes for enzyme immunoassays (EIA) because they lose little activity by chemical modification, and because of their high sensitivity, high stability and wide selection of colorimetric, fluorometric, and chemiluminometric substrates. Peroxidase-labeled protein is generally prepared using a reactive periodate-treated peroxidase. An aldehyde group of peroxidase derived from a sugar chain by the periodate oxidation reaction forms Schiff base with an amine group of the protein to create a covalent bond between the protein and peroxidase, After the conjugation reaction, a reducing agent is usually required to convert the Schiffs base to a C-N single bond, stabilizing the peroxidase conjugate. Dojindo’s Peroxidase Labeling Kit- NH2 and Alkaline Phosphatase Labeling Kit- NH2 can be used to simply and rapidly label both macromolecules (Fig. 5) and small molecules with amino groups. Conjugates prepared with this kit have higher enzyme activity and are more stable than other commercially available peroxidase or alkaline phosphatase-labeled antibodies or streptavidins. Peroxidase Labeling Kit-SH and Alkaline Phosphatase Labeling Kit-SH are used for labeling molecules with sulfhydryl groups. Except for the reduction of protein before labeling SH groups, the overall procedures for labeling are the same for both kits. Because of site-specific labeling with Kit-SH, enzyme-labeled IgGs prepared using Labeling Kit-SH sometimes yield higher sensitivity than conjugates prepared by Labeling Kit-NH2. Peroxidase Labeling Kits and Alkaline Phosphatase Labeling Kits are used for labeling small molecules as well as macromolecules. Small molecules with amino group(s) or sulfhydryl group(s) can be labeled with Labeling Kit- NH2 and Labeling Kit-SH, respectively. The labeling process is indicated in Fig. 4. The average number of small molecules conjugated to an enzyme is 2 per enzyme molecule. The conjugate can be used for competitive ELISA, receptor research, and other assays.
Fluorophore Labeling Kits
Fluorescent compounds such as fluorescein, rhodamine, cyanine dyes, and phycobiliproteins are commonly used for labeling biological molecules, especially proteins and DNA. Detection methods using these fluorescent materials are routine and well established. However, labeling conditions must be individually determined depending on the type of molecule, reactivity of the labeling agents, and the type of functional group to be labeled. Sometimes, pre-treatment of the sample may be necessary to remove molecules and reagents that interfere with the labeling reaction. Fluorophore Labeling Kits, such as Phycoerythrin, Allophycocyanin, Fluorescein, Rhodamine, and HiLyte dyes, are for labeling macromolecules with amino groups or sulfhydryl groups. Labeling processes of Kit-NH2 and Kit-SH are indicated in Fig. 6. The fluorescent compounds in these kits are water soluble and each vial of fluorescent compound can prepare a labeled IgG with 4 to 7 fluorescent molecules. On average, there are 1 to 2 phycobiliprotins per IgG. These kits include a purification system for the proteins to be labeled so that even protein solutions containing materials that interfere with the reaction, such as sodium azide, Tris buffer, and thiols, can be used with these kits. The fluorescent spectra of these reagents are shown on page 79.
|
|
|
|
Fig. 5 Enzyme Labeling Kit labeling process |
Fig. 6 Fluorophore or Biotin Labeling Kit labeling process |
Biotin Labeling Kits and Reagents
Peroxidase-labeled streptavidin and alkaline phosphataselabeled streptavidin are used for the detection of biotin-tagged macromolecules in EIA. Several types of biotin labeling reagents, such as amine reactive biotins, sulfhydryl reactive biotins, and aldehyde reactive biotins, are available. Spacers between the biotin molecule and the reactive group are also available in various lengths. Biotin Labeling Kit- NH2 is a ready-to-use kit for labeling amino groups of proteins. Because there is a longer spacer between the biotin and succinimidyl ester (NHS) in this kit’s biotin labeling reagent, the binding ability of streptavidin with a conjugated biotin molecule is almost identical to that of a free biotin molecule. Biotin Labeling Kit-SH uses Biotin-PE maleimide for the biotin labeling of sulfhydryl groups. Labeling processes of Kit- NH2 and Kit-SH are indicated in Fig. 6.
Rapid Labeling Kit series
Ab-10 Rapid Labeling series are specialized for 10 μg antibody labeling with a quick and simple manner. R-Phycoerythrin, peroxidase and fluorescein are available. Each conjugate can be prepared in 30 mins.
IgG Purification Kits
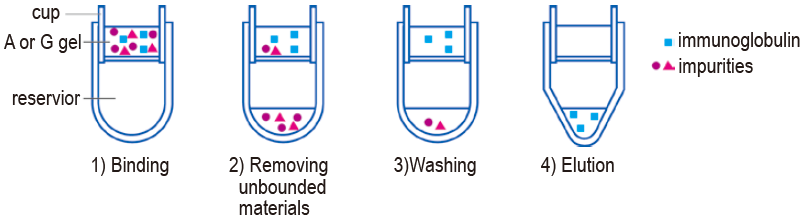
Commercially available antibody solutions sometimes contain a stabilizer, such as bovine serum albumin or gelatin, which interfere with labeling reaction. Dojindo’s IgG Purification Kits are useful for preparing a small amount of a purified antibody that can be applied directly to Dojindo’s labeling kits. IgG Purification Kit-A and Kit-G contain protein-A and protein-G coated silica gel, respectively. In using these kits there are four steps in purifying IgG: 1) binding IgG to the gel, 2) removing unbounded materials, 3) washing, and 4) recovery of IgG from gel. Most IgG molecules bind to protein A and protein G in 2 minutes. Because all solution is filtered from silica gel by centrifuging, the sample solution can be recovered without dilution. If the first step in the purification process does not sufficiently recover IgG, the same sample solution can be processed once more to recover additional IgG. Protein A gel and Protein G gel in the kit are capable of isolating IgG from serum with 80 to 90% purity. The gels can be used at least 20 times without diminishing IgG recovery rate.
|
Fig. 7 IgG Purification process |
Chelate Labeling Reagents
Chelate labeling reagents are used to bind metal ions to macromolecules. Macromolecules labeled with radioactive metal ions such as 99Tc and 111In are used for radioimaging with a scintillation camera to investigate macromolecule distribution in the body. Meares’ reagents are chelate-labeling reagents that have an EDTA structure and a reactive functional group such as isothiocyanate, maleimide, and bromoacetamide. Bromoacetamide-type Meares’ reagent, called “BABE” (4-bromoacetamidobenzyl-EDTA), is used for the analysis of proteinprotein or protein-DNA interaction sites.
Bifunctional Cross-Linking Reagents
Cross-linking reagents are used for the conjugation of two or more macromolecules. Cross-linking reagents are classified into two major groups, hetero-bifunctional and homobifunctional. Heterobifunctional cross-linking reagents have two types of reactive groups in their molecules, succinimide and maleimide. These two types of reactive groups make it possible, through modification by hetero-bifunctional reagents, for the biological materials to perform different functions, such as amine reactive and thiol reactive function. These modified materials may then react with other materials through the attached functional groups. In general, conjugated enzymes and monoclonal antibodies for enzyme immunoassay (EIA) are prepared using these heterobifunctional cross-linking reagents. Of the reagents that have succinimide and maleimide as their functional groups, aliphatic compounds such as GMBS and EMCS are superior to aromatic compounds in the stability of maleimide in basic conditions. Four types of maleimide/succinimide cross-linking reagents are available for conjugation, which alters the length of the space between an enzyme and an antibody. Water-soluble cross-linking reagents are also available. Water-soluble cross-linking reagents do not require organic solvents such as DMF or DMSO to prepare a working solution. Therefore, these water-soluble cross-linking agents are useful for proteins sensitive to organic solvents.
Spectra Data
Fig. 8 indicate excitation spectra and emission spectra of IgG conjugates prepared by Fluorophore Labeling Kit- NH2. The maximum wavelength of the conjugate prepared by Fluorophore Labeling Kit-SH is almost identical to that of the corresponding conjugate prepared by Fluorophore Labeling Kit- NH2.
|
Fig. 8 Emission spectra of Fluorphore conjugated IgG |
Product
- Protein Labeling Kits
-
Code Product name Unit size AP01 Protein Purification Kit
IgG Purification Kit - A1 set AP02 Protein Purification Kit
IgG Purification Kit - G1 set LK01 Antibody / Protein Labeling
Fluorescein Labeling Kit - NH23 samples LK03 Antibody / Protein Labeling
Biotin Labeling Kit - NH23 samples LK09 Antibody / Protein Labeling
Peroxidase Labeling Kit - SH3 samples LK10 Antibody / Protein Labeling
Biotin Labeling Kit - SH3 samples LK11 Antibody / Protein Labeling
Peroxidase Labeling Kit - NH23 samples LK12 Antibody / Protein Labeling
Alkaline Phosphatase Labeling Kit - NH23 samples LK13 Antibody / Protein Labeling
Alkaline Phosphatase Labeling Kit - SH3 samples LK14 Antibody / Protein Labeling
HiLyte Fluor™ 555 Labeling Kit - NH23 samples LK15 Antibody / Protein Labeling
HiLyte Fluor™ 647 Labeling Kit - NH23 samples LK21 Antibody / Protein Labeling
Allophycocyanin Labeling Kit - NH23 samples LK23 Antibody / Protein Labeling
R-Phycoerythrin Labeling Kit - NH23 samples LK24 Antibody / Protein Labeling
Allophycocyanin Labeling Kit - SH3 samples LK26 Antibody / Protein Labeling
R-Phycoerythrin Labeling Kit - SH3 samples LK31 Antibody / Protein Labeling
ICG Labeling Kit - NH23 samples LK32 Antibody Labeling
Ab-10 Rapid Fluorescein Labeling Kit3 samples LK33 Antibody Labeling
Ab-10 Rapid Peroxidase Labeling Kit3 samples LK34 Antibody Labeling
Ab-10 Rapid R-Phycoerythrin Labeling Kit3 samples LK35 Antibody Labeling
Ab-10 Rapid HiLyte Fluor™ 555 Labeling Kit3 samples LK36 Antibody Labeling
Ab-10 Rapid HiLyte Fluor™ 647 Labeling Kit3 samples LK37 Antibody Labeling
Ab-10 Rapid Biotin Labeling Kit3 samples LK51 Antibody / Protein Labeling
Peroxidase Labeling Kit - NH2 (for 1mg)1 sample LK55 Protein Labeling Kit
Biotin Labeling Kit - NH2 (for 1mg)1 sample - Protein Labeling Reagents
-
Code Product name Unit size C309 Fluorescent Dye for Cytoplasmic Staining
CFSE10 mg I254 Fluorescent Labeling Reagent
ICG-Sulfo-OSu1 mg I271 Fluorescent Labeling Reagent
IC3-OSu special packaging20 μg x3 I272 Fluorescent Labeling Reagent
IC5-OSu special packaging20 μg x3 - HPLC Derivertization Reagents
- Biotion Labeling Reagents
-
Code Product name Unit size B299 Biotin Labeling Reagent
Biotin-PEAC5-maleimide10 mg B304 Biotin Labeling Reagent
Biotin-OSu10 mg B305 Biotin Labeling Reagent
Biotin-AC5-OSu10 mg B306 Biotin Labeling Reagent
Biotin-(AC5)2-OSu10 mg B319 Biotin Labeling Reagent
Biotin Sulfo-OSu10 mg B320 Biotin Labeling Reagent
Biotin-AC5 Sulfo-OSu10 mg B321 Biotin Labeling Reagent
Biotin-(AC5)2 Sulfo-OSu10 mg B572 Biotin Labeling Reagent
Biotin-SS-Sulfo-OSu100 mg B592 Biotin Labeling Reagent
Biotin-PE-maleimide10 mg - Related Reagents
-
Code Product name Unit size C554 Substrate for Immunofluorescence
CLAMP F405-Signal Boosting10 µl D033 Metal Chelate
DTPA anhydride1 g
5 gS291 Cross-Linker
SPDP100 mg - Exsosome Labeling
-
Code Product name Unit size EX01 Exosome Membrane Fluorescent Staining
ExoSparkler Exosome Membrane Labeling Kit-Green5 samples※ EX02 Exosome Membrane Fluorescent Staining
ExoSparkler Exosome Membrane Labeling Kit-Red5 samples EX03 Exosome Membrane Fluorescent Staining
ExoSparkler Exosome Membrane Labeling Kit-Deep Red5 samples EX04 Exosome Protein Fluorescent Staining
ExoSparkler Exosome Protein Labeling Kit-Green5 samples EX05 Exosome Protein Fluorescent Staining
ExoSparkler Exosome Protein Labeling Kit-Red5 samples EX06 Exosome Protein Fluorescent Staining
ExoSparkler Exosome Protein Labeling Kit-Deep Red5 samples EX10 Exosome Isolation Kit
ExoIsolator Exosome Isolation Kit3 tests EX11 Filter for exosome isolation kit
ExoIsolator Isolation Filter10 pieces